Context
BD sought to structure a unified clinical intelligence infrastructure enabling pharmacists, analysts, and researchers to investigate safety, utilization, and cost signals across fragmented healthcare datasets.
The challenge was not data availability, but investigative architecture. Analysts needed to move fluidly between macro safety signals, cohort comparisons, financial exposure trends, and patient-level patterns without interpretive fragmentation. The existing system supported reporting. BD required a signal-oriented investigative model.
This engagement focused on restructuring the platform from static reporting logic into an analytical intelligence environment capable of supporting hypothesis formation, longitudinal exploration, and role-adaptive interpretation.
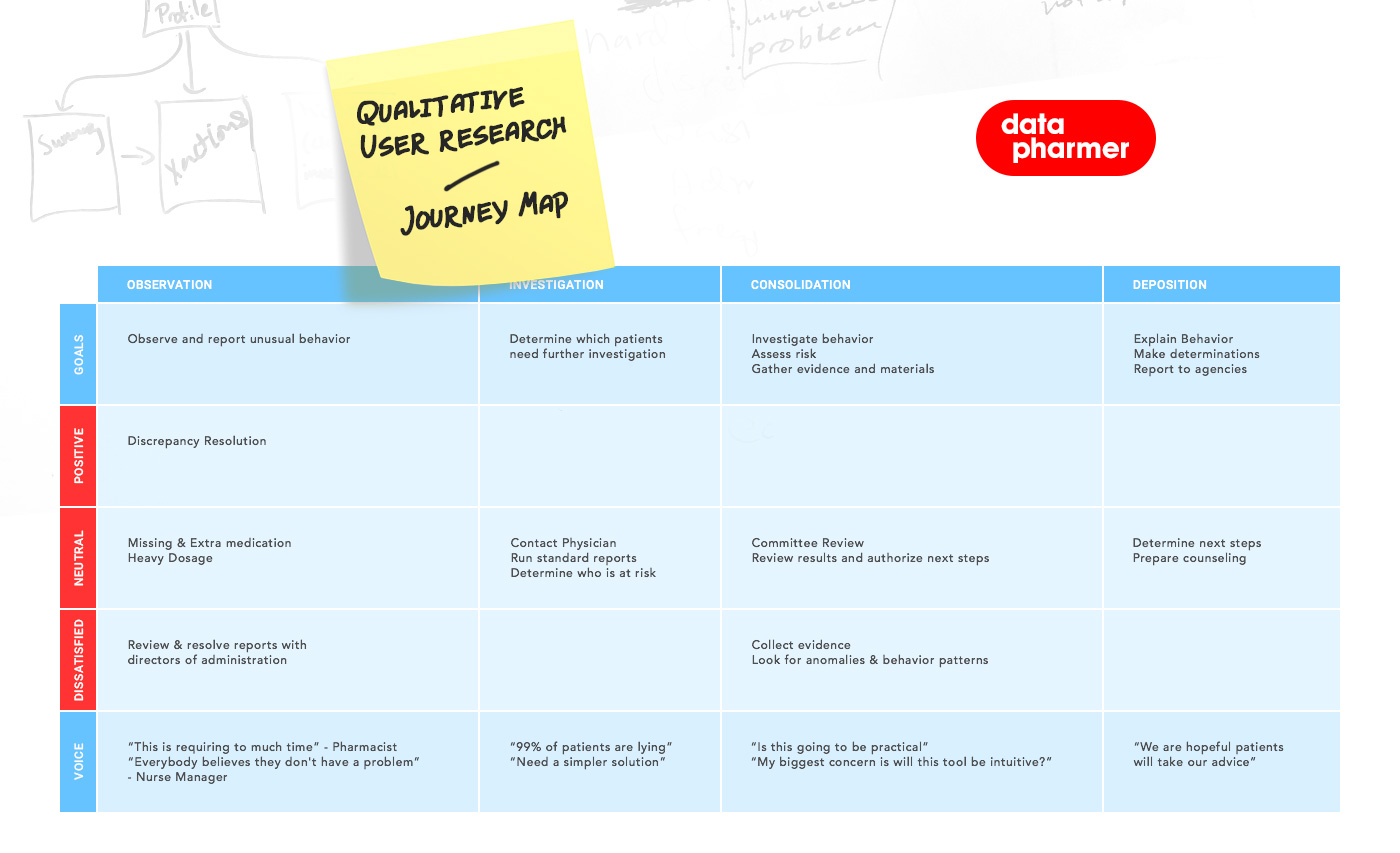
Research focused on mapping investigative flows across roles, identifying structural friction, and exposing where signal density exceeded interpretive clarity. Workflow tracing and system mapping clarified how pharmacists, analysts, and researchers navigated safety, utilization, and cost dimensions differently — and where the infrastructure needed modular restructuring.
Challenge
Researchers, pharmacy analysts, and clinical stakeholders approached the platform with fundamentally different investigative intentions. Some entered the system to examine emerging safety patterns. Others needed to compare treatment cohorts across cost and utilization variables. Still others were focused on longitudinal adherence trends or outcome variability across patient populations.
The existing product environment supported reporting, but it did not fully support open-ended investigation. Users could retrieve data, but forming and refining hypotheses required workarounds, manual cross-referencing, and navigation across disconnected views.
The opportunity was to shift from static report logic to investigative logic — enabling users to begin with a signal and move fluidly across timeframes, cohorts, treatment categories, and financial impact without breaking cognitive continuity. The system needed to support structured inquiry rather than linear consumption.
My Role
I led the research, synthesis, workflow modeling, and UX architecture across the product surface. Responsibilities included user interviews, persona modeling, workflow mapping, information hierarchy development, wireframing, prototyping, and interaction design.
Beyond interface design, the engagement focused on clarifying how different stakeholder roles approached investigation and restructuring the product’s logic accordingly.
Structural Insight
Research revealed that investigation patterns rarely followed predefined report flows. Users typically began with a high-level anomaly or macro signal — a cost spike, safety deviation, utilization shift, or unexpected treatment pattern — and then drilled into adjacent variables to understand correlation and cause.
What became clear was that the friction was not a lack of data. It was the absence of an integrated interpretive layer. Each stakeholder role interpreted the same dataset differently: executives looked for systemic exposure, analysts required multi-variable comparison and filtering flexibility, pharmacists prioritized interaction and safety clarity, and researchers focused on longitudinal outcomes across cohorts.
The platform therefore required a modular intelligence architecture built on a shared analytical backbone. Insight surfaces needed to adapt to investigative intent while preserving structural coherence across clinical, operational, and financial dimensions. The redesign moved the system away from rigid reporting templates and toward a role-aware, signal-oriented exploration model.
Product Architecture
The shipped product structured investigation around flexible filtering models, cohort comparison views, layered visualizations, and contextual drill-down pathways. Information hierarchy was simplified to prioritize query clarity before visualization complexity.
Wireframes translated research findings into navigational logic that reduced cognitive switching and improved interpretability across roles.
Later in the engagement, exploratory prototypes introduced a more elevated visual system and tighter signal clustering models aimed at improving comparative clarity and longitudinal pattern recognition. While not fully realized before contract conclusion, this direction informed the future evolution of the platform.
Investigation Architecture
Directional Evolution
Early-stage prototypes explored enhanced clustering logic, comparative signal grouping, and cleaner visual hierarchy models to improve investigative speed and longitudinal interpretation. While not fully implemented before engagement conclusion, these explorations defined the product’s next evolution.
Validation
User testing focused on interpretability, interaction friction, and investigative continuity. Sessions revealed where filtering logic created bottlenecks and where visual density hindered insight formation.
Iteration reduced interface complexity, clarified interaction affordances, and improved time-to-interpretation during investigative tasks.
Outcome
The engagement repositioned Data Pharmer from a report-oriented application into a structured clinical intelligence infrastructure. By reorganizing investigative logic, clarifying signal hierarchy, and modularizing analytical surfaces, the platform moved closer to supporting scalable inquiry across safety, utilization, and economic dimensions.
The shipped system reduced structural fragmentation, improved interpretive continuity across roles, and established an analytical backbone capable of supporting expanding data complexity.
Exploratory architectural work conducted during the later phase of the engagement defined a forward trajectory for the platform’s next stage of maturity — including enhanced signal clustering and longitudinal modeling capabilities.
The result was not a visual refresh. It was an architectural shift toward scalable investigative intelligence inside a global healthcare organization.